At Rush, orthopedic surgeons do more than repair joints. They restore lives. Whether helping a weekend warrior return to the trail or a professional athlete get back on the field, the goal is the same: reduce pain, improve mobility and help people do what they love.
But healing isn’t just physical. Our neurological health matters, too.
“Pain, fear and anxiety have a huge impact on the rehabilitation process,” explained Brian Forsythe, MD, the Tim and Keli Walbert Endowed Chair in Orthopedic Surgery. “They delay muscle activation, limit range of motion and restrain overall movement.”
These feelings cause you to adapt your movements, interfering with the brain’s signals, slowing recovery and increasing the risk of reinjury. Fear and anxiety can lead to arthrogenic muscle inhibition, which makes it hard to contract muscles involved in movement, and even a clinical fear of movement, called kinesiophobia. While we don’t yet fully understand the roles fear and anxiety play in the rehabilitation process, Forsythe and his colleagues are changing that.
To generate a baseline understanding of how psychological factors like kinesiophobia might affect recovery, Forsythe and his collaborators conducted an analysis of return-to-play and performance metrics for Major League Soccer players recovering from ACL injuries.
The team’s study revealed that despite faster return-to-play times over the last decade, many athletes struggled to reach their pre-injury performance levels.
Defenders returned to their preinjury performance level after two years, while midfielders scored more goals by their third year post-ACL reconstruction. Forwards, though, whose roles demand explosive movement and agility, experienced decreases in goals scored, minutes played and shots on goal into year four.
“On the one hand, these findings may reflect accelerated rehab protocols and possibly improved surgical techniques,” Forsythe said. “At the same time, they suggest physical healing alone is not enough.”
That insight is driving a new direction in rehabilitation to accelerate recovery. Forsythe and his team are building on this study by exploring how neurofeedback visualization technology can help retrain the brain, expedite muscle reactivation and decrease arthrogenic muscle inhibition.
Enhancing rehabilitation using technology
Five years ago, Forsythe was introduced to a neuroscience-based visualization tool used by elite athletes to improve performance. He immediately wondered if this technology could support a more efficient rehab process and promote healing.
“When I met the developers of the technology five years ago, they didn't have a rehab platform,” he recalled. “The technology is a cool device in sports performance, but as a clinician-scientist who takes care of recreational and professional athletes when they get hurt, I saw an even greater need for this technology in the rehab process.”
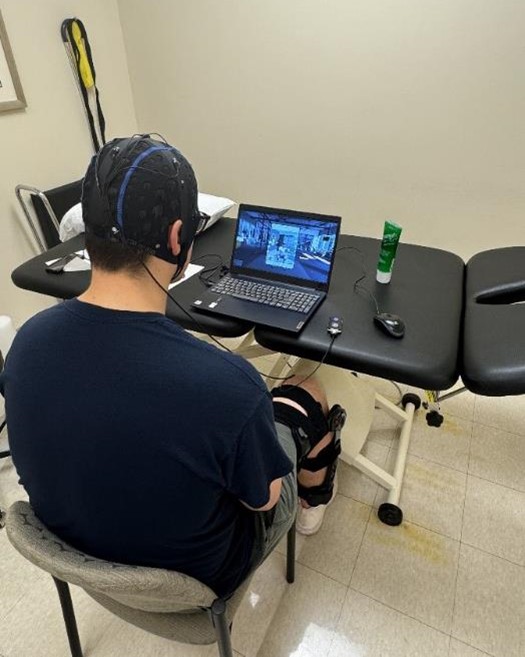
In collaboration with colleagues across the department, the Rush Motion Analysis Lab and Midwest Orthopaedics at Rush physical therapy clinics, Forsythe recently launched a randomized control trial to determine if the neurofeedback visualization training makes a measurable difference for patients. The complex study took over a year to plan.
“There are a lot of moving parts to conduct a study like this one,” Forsythe said. “It requires a high level of organization, strategy and enthusiasm to execute it.”
Patients who have undergone an ACL reconstruction, femoroacetabular impingement surgery, hip labral repair, or a total hip or knee replacement are eligible to enroll in the study. They will be randomly assigned to either the study or control group. The study group will participate in neurofeedback visualization training for eight weeks following their surgery alongside a standard physical therapy regimen. In the Motion Analysis Lab, the team will learn how well patients’ muscles are contracting, how strong they are and how much energy they generate.
“If we can adapt this technology for rehab, we could make a profound impact on the recovery process and even decrease the odds of reinjury,” Forsythe said.
How philanthropy puts us on the path to progress
Private support is essential to advance orthopedic medicine. Funding from the National Institutes of Health has led to tremendous strides in orthopedic care, but only 2% to 3% of its budget funds research into musculoskeletal diseases, while half of Americans experience them. Philanthropy fills the gap.
Gifts like those from Tim and Keli Walbert keep Rush at the forefront of innovation and care. Their recent, historic investment in the department will advance opportunities for early-career orthopedic researchers and fund discoveries to bring pain relief to millions of Americans. The gift established four endowed funds, including the Tim and Keli Walbert Endowed Chair in Orthopedic Surgery, which honors Forsythe’s groundbreaking research to find better treatment options for bone and joint pain, including the neurofeedback visualization training study.
“The Walberts’ investment in our orthopedic research program has allowed me to assemble a team of researchers and experts who can execute a complex study like this one,” Forsythe said.
Because studies like the neurofeedback visualization trial inform and influence orthopedic practice nationally and internationally, philanthropic gifts have a ripple effect, reaching patients beyond Chicago. It allows the team to build on their findings and apply them to other areas, like shoulder surgeries, where there is a great need for more evidence-based therapy protocols.
“The translational nature of this research will make a profound difference, touching thousands, if not millions, of lives,” Forsythe said.




